Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
千葉 薫*; 松井 拓郎*; 茶竹 俊行*; 大原 高志; 田中 伊知朗*; 油谷 克英*; 新村 信雄*
Protein Science, 32(10), p.e4765_1 - e4765_13, 2023/10
被引用回数:0 パーセンタイル:0(Biochemistry & Molecular Biology)In structural biology, peptide bonds, fundamental linkages between hundreds of amino acids, of which a protein molecule is composed, have been commonly treated as a plane structure just as Linus Pauling et al. proposed. In this paper, a site-specific peptide bond relaxation mechanism by deuterons whose localization has been suggested by neutron crystallography is proposed. A comprehensive study using X-ray and neutron diffraction and 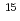 N chemical shifts of individual amide nitrogen atoms within the same peptide bond strongly suggests the relaxation of the electronic resonance structure because of site-specific modulation by protons/deuterons localized on the electron orbital of the carbonyl oxygen.
N chemical shifts of individual amide nitrogen atoms within the same peptide bond strongly suggests the relaxation of the electronic resonance structure because of site-specific modulation by protons/deuterons localized on the electron orbital of the carbonyl oxygen.